|
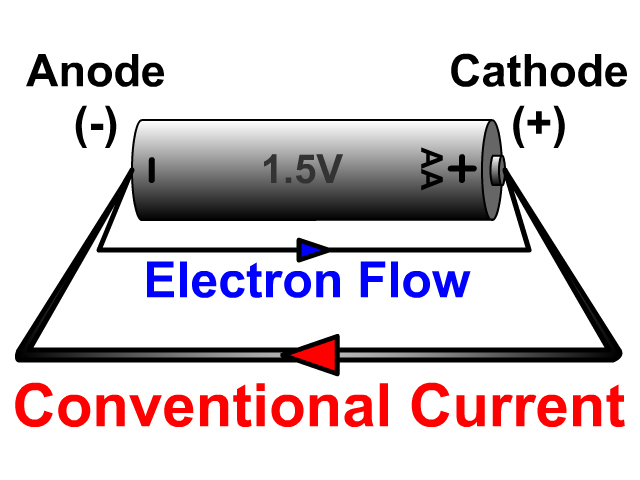
It allows electrolytic current to flow across it, but keeps the reactive components (Zn and MnO 2) separated so they do not react directly. The "porous separator" is simply a paper-like material that provides the functionality of a salt bridge. The "current collector" in this case acts just like an inert electrode and simply allows for electrical contact with the zinc anode paste. In powder form, the zinc has more surface area for electron transfer which allows for higher electric currents. The zinc anode in modern batteries is a compressed Zn powder mixed with aqueous KOH (another paste). THe graphite helps ensure better electrical contact throughout the compressed powder/paste and is not shown in the shorthand notation. The MnO 2 is a powder and is mixed with graphite and aqueous KOH to make a paste. Note that the positive terminal in the diagram is just the contact point of a stainless stell can that is in electrical contact with the active cathode material of MnO 2. The diagram shown here is a cut-away diagram of a typical alkaline cell. Adjusting for the actual concentrations used in the manufacture of the batteries (using the Nernst equation), you get a battery with somewhere between 1.50 and 1.65 V. Notice that the overall standard cell potential for this cell will be +1.43 V. The graphite then acts as an extended electrode surface in contact with all the active MnO 2. MnO 2 is a non-conductive oxide so in order to make electrical contact, graphite powder is mixed with the MnO 2 to make a paste. The cathode active material (what gets reduced) is MnO 2(s). All of these cells share the same exact chemistry. "Letter" meaning the D-cell, C-cell, AA-cell, and AAA-cell. Certainly the most popular shape is the familiar cylindrical shape of the "letter" cells. These are typically cells that have a voltage of 1.5 V. Dry Cell, Zn-Carbon, LeClanche Cell, Heavy DutyĪlkaline Cells: Probably the most familiar of batteries is the alkaline cell.Primary cells are meant to be used once and then discarded. The reverse reaction does not occur efficiently enough to allow one to regenerate the reactants from the products. The chemical reactions (and side reactions) in primary cells really only go in one direction and that is from reactants to products. This means a primary cell is a non-chargeable cell. 
The primary cell is a voltaic cell (battery) that is made to be a one-time use battery.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
